Investment
Investment Process
Ellington Healthcare Partners I, L.P. which invests in new and innovative opportunities in the use of Adult Stem Cells for regenerative medicine and other Stem Cell-based therapies and in Nanotechnology for its potential for enhanced drug delivery and other therapeutic and diagnostic applications. In this effort to find those investment opportunities that possess the highest degree of potential success, the managers of Ellington intend to follow a highly disciplined and rigorous review process.
Fund Management brings together three teams that are integrated in the review process that all potential investments must traverse: 1) world class scientists who have a deep knowledge of the state of science in the targeted investment area; 2) pharma industry experts with the experience necessary for market sizing, assessment of regulatory complexities and competitive analysis; and 3) experienced executives with proven investment and management skills for deal structuring and valuation. The following flow chart illustrates the process:
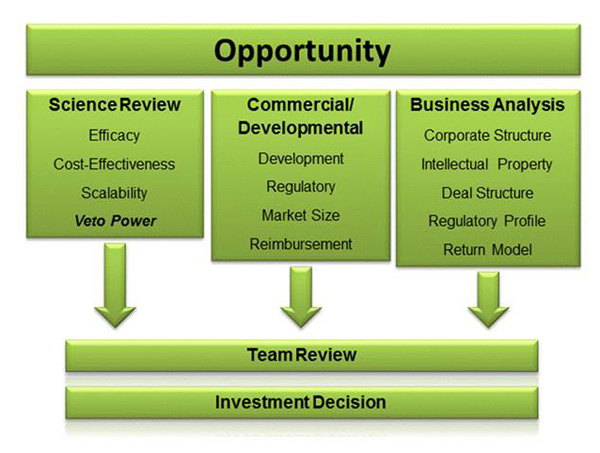
All potential investments will first be subject to an assessment and determination that the proposed investment has commercial viability. In this phase of the due diligence process, attention will be focused on the potential market for the therapy. Factors considered include: the incidence and prevalence of the targeted disease, the size of the population targeted by the therapy, the current standard of care and the potential improvement and/or economic advantage that the new therapy will have. The Commercial and Business Development Partners will also seek to determine the commercial potential of this new therapy relative to products that are currently in clinical development. With the increasing importance of managed care in the US market, it is also vital to judge the potential for this therapy with this audience and evaluate its potential to qualify for insurance reimbursement and its expected worldwide pricing parameters.
Only after passing the Commercial and Business Development Partners review will investment opportunities be considered for further investigation by the Fund’s Scientific Team Partners. In this phase of the due diligence process, attention will be paid to the validity of the science underlying the opportunity, trial design and objectives.
Once it is determined that the proposed investment has commercial viability and passes the scrutiny of the Fund’s Scientific Team Partners the next stage of due diligence will be performed by the Investment and Operations Team Partners. As part of this process, Fund Management will analyze a company’s operational and management structure, legal structure, including the protection and/or exposure of the company’s intellectual property, personnel, insurance and risk protection required, internal cash-flow forecasts and existing financial reporting systems.
Those prospective investment opportunities that successfully clear all three review stages of Fund Management will be candidates for a term sheet. The terms of investment must be approved by the Investment Committee. Once a successful closing is accomplished, one member of the Scientific Team and one additional member of the Management Team will be assigned to the portfolio company on an ongoing basis, including participation on the company’s Board of Directors.
EHP Investment Criteria
 The selection of Fund investments will be governed by the following investment criteria:
The selection of Fund investments will be governed by the following investment criteria:
- Breakthrough Product
- Sound Intellectual Property Protection
- Clearly definable milestones for development
- Sustainable technological advantage
- Market Size
- Foreseeable exit potential
- Above average business potential
- Scientist’s commitment and dedication
- Geography
Investment Focus: Regenerative Medicine
- It is well-established that Adult Stem Cells are clinically useful, providing remarkable regenerative and tissue-sparing properties
- Adult Stem Cells have a variety of clinical applications, including the ability to:
- Generate needed cells (e.g., blood)
- Promote the growth of tissue-resident cells
- Support native processes leading to tissue repair
- Dampen the inflammatory response to tissue injury – a major impediment to rapid tissue healing
- Correct genetic errors (via gene therapy)
Investment Focus: Nanotechnology

Nanotechnology holds great promise in optimizing pharmaceutical efficacy, delivery and safety and has wide applications in oncology and infectious disease
Nano-scale materials (Nanomedicine has tremendous direct clinical applicability and commercial potential:
- Diagnostics & Imaging
- Treatment
- Disease prevention
Investment Focus: Oncology
 Novel cancer therapies
Novel cancer therapies- Immunotherapy
- New chemotherapies
- Targeted therapy
- Management of adverse consequences
- Quality of life improvement.
